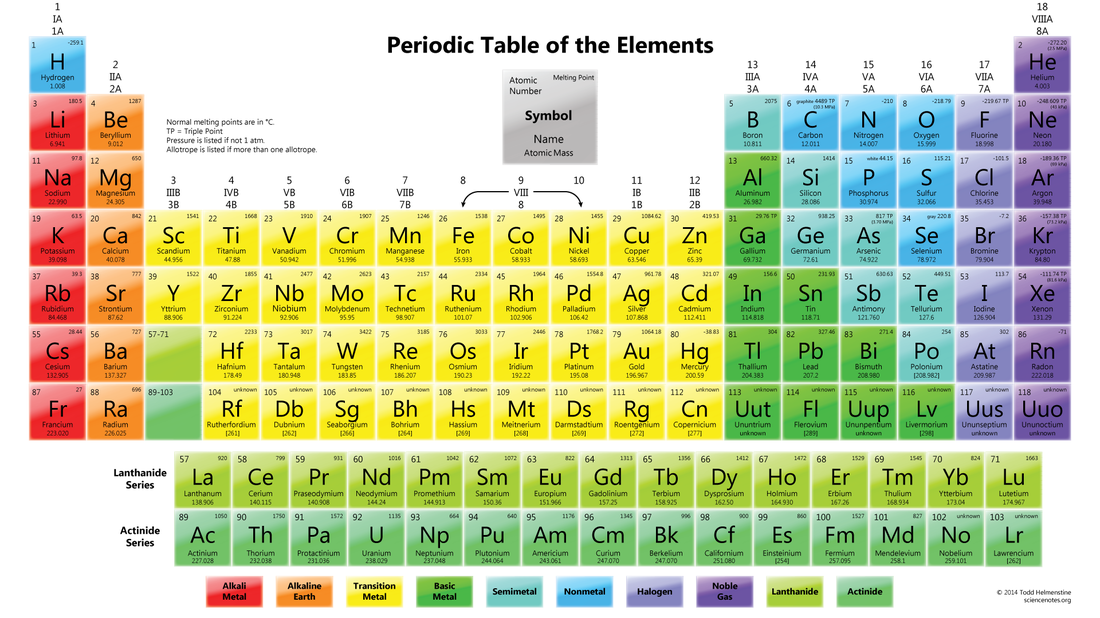
PERIODS
Every element in the top row (first period) has one orbitals for it electrons
GROUPS/FAMILIES
VALENCE ELECTRONS:
- Periods go left to right
- All of the elements in a period have the same number of atomic orbital
Every element in the top row (first period) has one orbitals for it electrons
GROUPS/FAMILIES
- Groups go up and down
- The elements in each group have the same number of electrons in the outer orbital (valence electrons)
VALENCE ELECTRONS: